 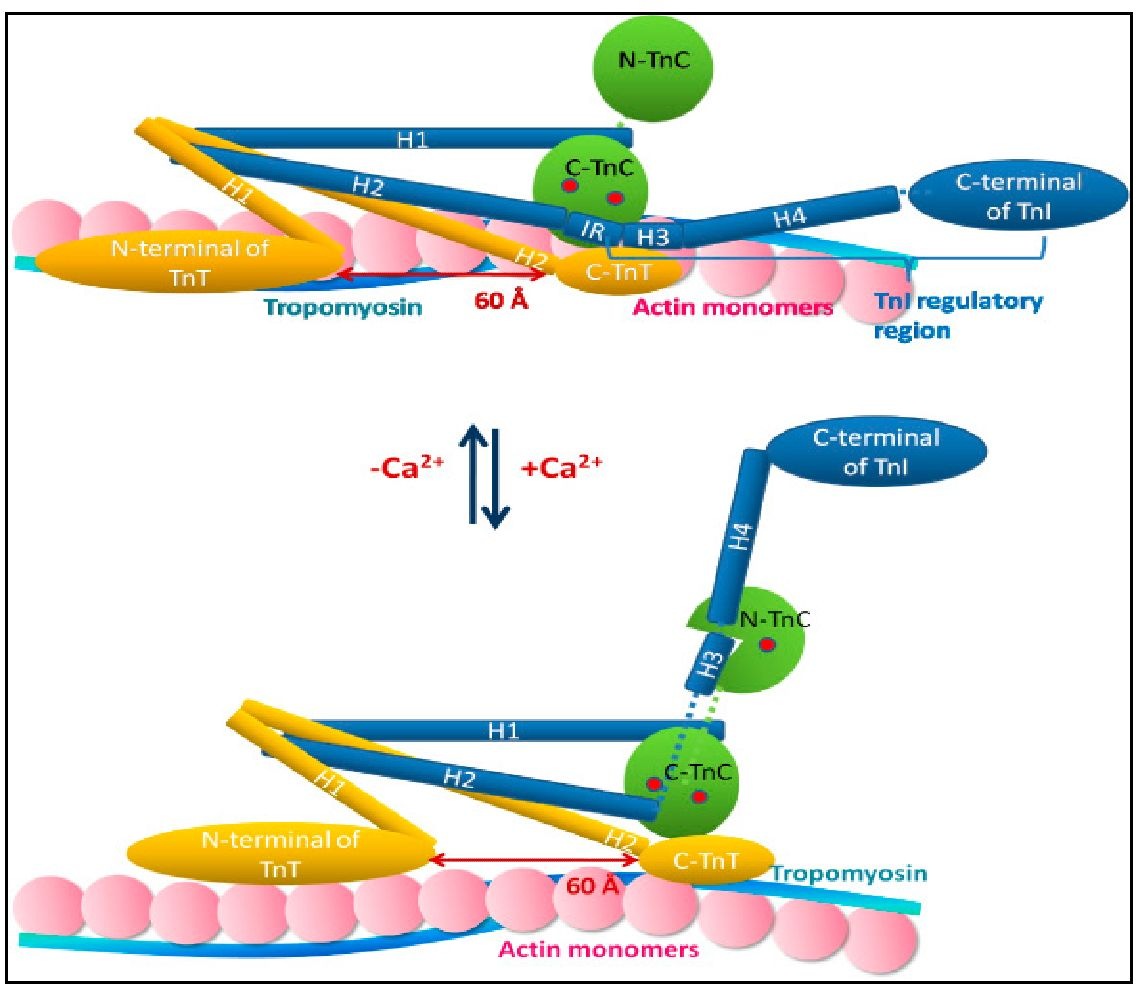
In all discordant cases the differences were minor and would have resulted in either no or minimal change in therapy. Sixty one (61) of 68 patient sample pairs collected by nurses showed acceptable agreement. The criteria for determining clinical equivalence of the iSTAT to the MDA II were (1) same clinical category (subtherapeutic INR3) or (2) paired values within ≤ 0.4 INR.įorty nine of 50 patient sample pairs collected by phlebotomists showed acceptable clinical agreement. We also obtained 68 near simultaneous capillary/venous test results for assessment of performance by non-laboratory personnel. We obtained simultaneous capillary/venous samples on 20 healthy volunteers for a normal range study and on 50 anticoagulated patients for a clinical evaluation. We report an evaluation of the iSTAT PT/INR with a comparison to the Tcoag MDA II analyzer. However, the performance of some devices has been shown to be suboptimal and there are only limited published data comparing specific devices to various central laboratory coagulation analyzers. For i-STAT test cartridge information and intended use, refer to individual product pages or the cartridge information (CTI/IFU) in the i-STAT Support area.Ībbott - A Leader in Rapid Point-of-Care Diagnostics.Point-of-care devices for performing a prothrombin time/international normalized ratio (PT/INR) using capillary blood samples are being increasingly used to monitor patients receiving anticoagulation therapy. Check with your local representative for availability in specific markets. Not all products are available in all regions. GDPR Statement | Declaration for California Compliance Law. Any person depicted in such photographs is a model. 
Photos displayed are for illustrative purposes only. Your use of this website and the information contained herein is subject to our Website Terms and Conditions and Privacy Policy. The products and information contained herewith may not be accessible in all countries, and Abbott takes no responsibility for such information which may not comply with local country legal process, regulation, registration and usage. This website is governed by applicable U.S. No use of any Abbott trademark, trade name, or trade dress in this site may be made without the prior written authorization of Abbott, except to identify the product or services of the company. Unless otherwise specified, all product and service names appearing in this Internet site are trademarks owned by or licensed to Abbott, its subsidiaries or affiliates. 
Analytical performance of the i-STAT cardiac troponin I assay. Apple FS, Murakami MM, Christenson RH, et al. Impact of point-of-care testing in the emergency department evaluation and treatment of patients with suspected acute coronary syndromes. ACC/AHA 2007 Guidelines for the Management of Patients With Unstable Angina/Non ST-Elevation Myocardial Infarction: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines for the Management of Patients With Unstable Angina/Non ST-Elevation Myocardial Infarction). 
2009 53:321- 328.ģ. Third Universal Definition of Myocardial Infarction (MI), Kristian Thygesen, Joseph S. A multicenter randomized controlled trial comparing central laboratory and point-of-care marker testing strategies: The disposition impacted by serial point of care markers in acute coronary syndromes (DISPO-ACS) trial. Irving, Tex: American College of Emergency Physicians April 2008.Ģ. Emergency Department Crowding: High-Impact Solutions. This page is only accessible to registered i-STAT customers.ġ. Reference ranges (sometimes referred to as normal ranges) in the default Customization profile can be found behind login on the i-STAT Cartridge and Test Information (CTI) Sheets/Instructions for Use (IFU) page. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
